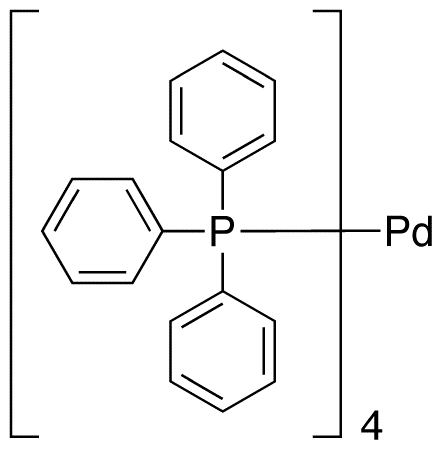
![14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・209-14643・203-14641[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation 14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・209-14643・203-14641[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation](https://labchem-wako.fujifilm.com/sc/01/14221-01-3.png)
14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・209-14643・203-14641[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation
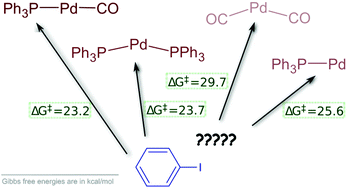
Viable pathways for the oxidative addition of iodobenzene to palladium(0)- triphenylphosphine-carbonyl complexes: a theoretical study - Dalton Transactions (RSC Publishing)
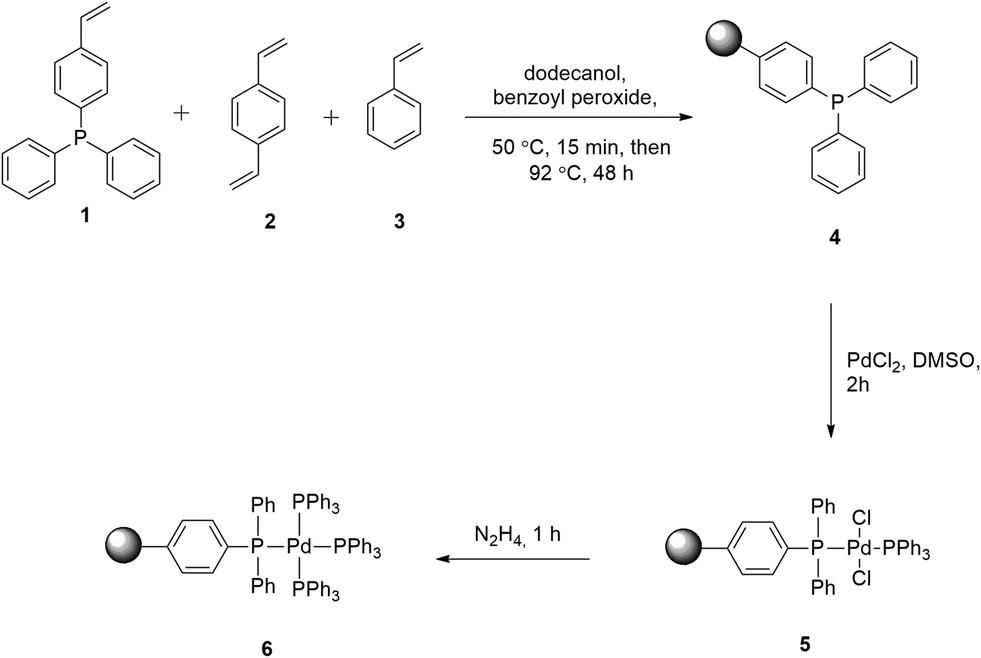
Immobilized tetrakis(triphenylphosphine)palladium(0) for Suzuki–Miyaura coupling reactions under flow conditions - Reaction Chemistry & Engineering (RSC Publishing)

Bis(triphenylphosphine)palladium(II) Dichloride 13965-03-2 | Tokyo Chemical Industry Co., Ltd.(APAC)

Tetrakis(triphenylphosphine)palladium(0), 99,8% (metals basis), Pd 9% min, Alfa Aesar™ 2g Tetrakis(triphenylphosphine)palladium(0), 99,8% (metals basis), Pd 9% min, Alfa Aesar™ | Fisher Scientific

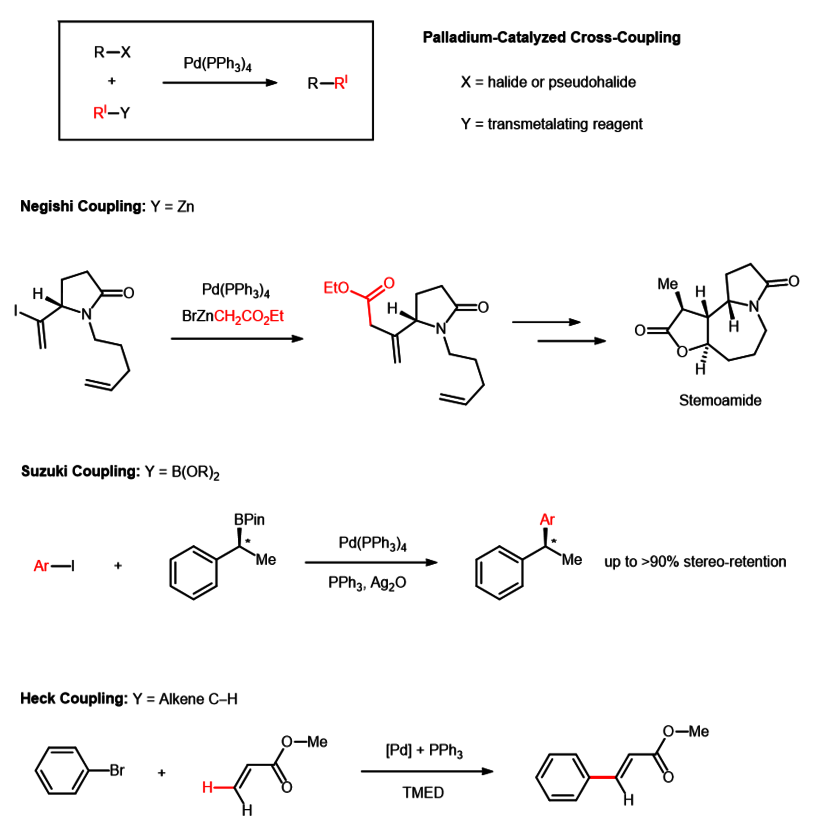
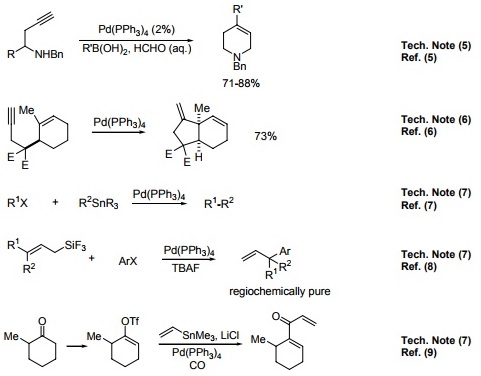
Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds




palladium(II)%C2%A0chloride.jpg)